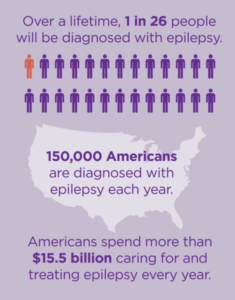
Epilepsy is the 4th most common neurological disorder affecting about 3 million Americans and 65 million people worldwide. Each year, 150,000 individuals in the US and another 2.4 million worldwide are diagnosed with epilepsy.
About 80% of individuals diagnosed with epilepsy are from low-to-middle income countries. These are nations where there is a higher risk for common diseases, more accidents involving brain injuries, and poor preventative healthcare.
Epilepsy can also be caused by congenital abnormalities, genetic predisposition, brain infections, brain cancer and tumors, and neurological disorders like stroke or injuries to the brain.
Medications can control epilepsy symptoms, but they may have serious long-term complications. Researchers looking for safer, more effective alternative epilepsy treatments have discovered that cannabinoids, particularly CBD, may be the answer.
Understanding Epilepsy
 Epilepsy is a chronic neurological disorder caused by abnormal brain cell activity, resulting in recurrent and unprovoked seizures. These seizures may involve only a part of the body and brain (partial seizures) or the whole body and both brain hemispheres (generalized seizures).
Epilepsy is a chronic neurological disorder caused by abnormal brain cell activity, resulting in recurrent and unprovoked seizures. These seizures may involve only a part of the body and brain (partial seizures) or the whole body and both brain hemispheres (generalized seizures).
Epilepsy symptoms vary depending upon which part of the brain is affected. Symptoms include convulsions, involuntary movements, temporary paralysis, loss of consciousness, incoordination, confusion, memory loss, mood changes, and other cognitive changes.
Pathophysiology of Epilepsy
The brain governs all bodily functions. Communication from the brain to various parts of the body is facilitated by excitatory neurotransmitters like glutamate, which are controlled by inhibitory neurotransmitters.
In the pathophysiology of epilepsy1, a neurotransmitter imbalance is caused by certain stimuli, such as gene abnormalities, brain infections or injuries, and metabolic abnormalities. This imbalance results in the neurons becoming hyperexcitable. Although one hyperexcitable neuron will not trigger a seizure, a group will. Neurons activate neural networks and neuron groups fire together. Seizures are triggered by the hyperexcitable neurons, eventually causing chronic inflammation which increases seizure severity and recurrence.
Research shows that brain inflammation2 also plays a role in epilepsy. During a seizure, cells activate to produce proinflammatory cytokines (molecules that stimulate the movement of cells towards sites of inflammation), which increases the production and release of other proinflammatory molecules, resulting in worsening chronic symptoms. Cytokines affect glutamate transporters3 and inhibit astrocytes4 (cells that regulate the transmission of electrical impulses within the brain) responsible for glutamate reuptake.
Inhibiting glutamate reuptake causes a higher glutamate concentration in the synapse and prompts the astrocytes to release more glutamate. Astrocytes exhaust glycogen stores trying to reabsorb this excess glutamate. This causes the transporters to work in reverse, thus releasing more glutamate into the synapses5. This catastrophic increase in glutamate leads to excitotoxicity.
Excessive glutamate also makes cells more permeable to calcium, which releases destructive enzymes damaging the mitochondria, producing reactive oxygen species (ROS) which releases more calcium, and eventually causes apoptosis (cell death). Overactivation of glutamate receptors is a key factor in developing epilepsy.
Current Treatments for Epilepsy
Anticonvulsant medications, which work by keeping the seizure from spreading, include:
- Diazepam – effective yet short-term anticonvulsant usually used in emergencies.
- Carbamazepine and phenobarbital – most common for partial and generalized seizures.
- Eslicarbazepine and oxcarbazepine – daily medications to help control seizures.
- Phenytoin – an anticonvulsant administered intravenously to control active seizures.
These epilepsy medications come with concerning side effects such as:
- Tiredness and fatigue
- Dizziness and headache
- Sleep disturbances
- Appetite loss
- Nausea and vomiting
- Vision changes
- Allergic reactions
- Problems walking and tremors
Long-term use of epilepsy medications can cause cognitive changes like irritability, increased anxiety, mood swings, aggression, paranoia, bone thinning, hair growth, liver and pancreas damage, blood clotting problems, and kidney problems.
Cannabis: An Alternative Treatment for Epilepsy
Many ancient societies throughout history have used cannabis to control seizures. However, because marijuana is classified as a Schedule I substance, research has been limited in the US. However, researchers in other countries (such as Israel) have been studying the plant for several decades. The discovery of the endocannabinoid system (ECS) in 1992 heightened interest in cannabis and its medicinal potential.
 The endocannabinoid system is a group of internal cannabinoid receptors located in the brain and throughout the central and peripheral nervous systems in all mammals. Its primary function is to assist the human body in maintaining homeostasis and balance while modulating various physiological functions. CB1 receptors, abundant in the central and peripheral nervous systems, are responsible for physiological functions involving mood, memory, appetite, and sleep. CB2 receptors, on the other hand, are found primarily in the immune system.
The endocannabinoid system is a group of internal cannabinoid receptors located in the brain and throughout the central and peripheral nervous systems in all mammals. Its primary function is to assist the human body in maintaining homeostasis and balance while modulating various physiological functions. CB1 receptors, abundant in the central and peripheral nervous systems, are responsible for physiological functions involving mood, memory, appetite, and sleep. CB2 receptors, on the other hand, are found primarily in the immune system.
The body produces two main endocannabinoids – anandamide, which binds primarily to CB1 receptors and 2-Arachidonoylglycerol, which binds to both CB1 and CB2 receptors.
Whereas endocannabinoids represent cannabinoids that are found in the human body, phytocannabinoids are cannabinoids that are present in some plant species, most notably the cannabis plant. There are more than 100 known cannabinoids. The two most well-known, and most abundant, are Tetrahydrocannabinol (THC) and Cannabidiol (CBD).
THC binds to CB1 receptors to produce therapeutic effects and create the psychoactive “high” associated with cannabis. CBD, which is non-psychoactive, has little affinity for the CB1 receptor, or the CB2 receptor for the matter. Instead, CBD operates as an indirect antagonist of the CB1 and CB2 receptor agonists. In layman’s terms, this means that CBD mutes the effects of all the things that would typically activate the CB1 and CB2 receptors. That is why, in addition to providing its own therapeutic benefits, CBD is known to counter the psychoactive effects of THC. It does this in a couple of ways:
- By binding with CB1 receptors’ allosteric sites6, CBD changes how THC binds with CB1 receptors resulting in a blockage of THC’s psychoactive effects.
- As a competitive inhibitor of THC, CBD prevents it from binding with a group of enzymes7 in the liver which breaks down THC into a simpler, more potent form.
Cannabinoids Increase Anandamide and Reduce Excitotoxicity
Hyperpolarization of the neuron’s membranes results in increased potassium conductance and decreased calcium conductance. Calcium plays a role in muscle contraction, and potassium plays a role in homeostasis. A high intracellular calcium concentration causes excitotoxicity and apoptosis. Low potassium levels cause muscle spasms and rigidity. The ECS modulates neuronal excitability. Excessive excitatory neurotransmitters produce anandamide to activate CB1 receptors, thereby reducing glutamate production and protecting the neurons from acute excitotoxicity8.
CBD, in particular, plays a significant role in this. CBD enhances anandamide9 and the neuroprotective effects against excitotoxicity by preventing fatty acid-binding proteins (FABP) from transporting anandamide to the enzyme which breaks it down. The result is more anandamide being available to activate CB1 receptors.
Cannabinoids Reduce Epileptiform Activities
THC activates CB1 receptors to control seizures. A study using magnesium discovered that THC was effective in inhibiting calcium spikes associated with decreased magnesium. Magnesium deficiency10 causes convulsions, tremors, increased psychotic behavior, and depression. With THC, the calcium spikes were reduced, so a CB1 antagonist was introduced, subsequently inhibiting THC’s antiepileptic effects and proving that the activation of CB1 receptors results in antiepileptic effects11.
CBD activates multiple channels to create its antiepileptic effects:
- Inhibits FABPs to increase anandamide level.
- Reduces calcium12 by modulating its release and uptake.
- Inhibits T-type calcium channels13, which control a cell’s excitability and modulate calcium influx.
CBD’s Neuroprotective Properties
Chronic brain inflammation, excessive excitatory neurotransmitters, and calcium influx into the cells cause epilepsy and excitotoxicity to worsen and increases ROS production and free radicals which cause cell death.
CBD is a powerful anti-inflammatory and a potent neuroprotective and antioxidant agent14. CBD helps control epilepsy symptoms in several ways:
- By influencing CB2 receptors, CBD suppresses the cytokine production, inhibits inflammation, and reduces glutamate in the synapses.
- CBD also influences calcium channels and mitochondria, preventing toxic calcium accumulation within the cells and reducing excitotoxicity.
- CBD affects glutamate receptors and suppresses glutamate transmission, decreasing excitotoxicity and neurotoxicity.
- Influencing the adenosine A1 receptors, CBD enhances their ability to modulate excitatory neurotransmitters like glutamate.
- CBD’s influence on glycine receptors enhances the receptor’s ability to modulate neuronal excitability.
- CBD helps increase anandamide levels.
Effectiveness of CBD as a Treatment for Epilepsy
There have been numerous human clinical studies that have demonstrated the efficacy of CBD as a treatment for epilepsy.
- In one study, 19 parents used CBD-enriched cannabis for their children who were suffering from epilepsy and were not responding to traditional treatment. 16 of the 19 parents surveyed stated that they saw improvement in their children’s seizure frequency15. Two children reported being completely seizure-free, eight reported an 80% reduction in seizure activity, and the remaining six children experienced a 25% to 60% decrease in seizure frequency. Parents also reported a significant improvement in their children’s other symptoms, including better sleep, increased alertness when awake, and improved moods.
- Another study involving 15 epilepsy patients proved that CBD is an effective antiepileptic drug16. During the study, eight patients were given CBD, and the rest received a placebo for four and a half months. All patients continued their traditional medications during this time. No participant reported adverse reactions, but four epilepsy patients reported being seizure-free with significant improvement in their symptoms, and 3 reported partial improvement. CBD was ineffective in only one patient.
- In a very recent example, children with severe childhood-onset epilepsy who haven’t responded well to antiepileptic medications took part in a study with their parents and physicians not knowing if their child is receiving Epidiolex (a CBD product) or placebo. The study showed that children receiving CBD had a 39% improvement in seizure frequency17, while those receiving a placebo showed 16% improvement.
Research on CBD and epilepsy18 shows promising results. Unlike THC, CBD has no psychoactive effects, yet provides similar therapeutic effects, making CBD an ideal cannabinoid-based treatment for epilepsy.
References:
1 David Y. Ko. July 12, 2016.
MedScape, Epilepsy and Seizures.
http://emedicine.medscape.com/article/1184846-overview
2 Annamaria Vezzani, et al. December 7, 2010.
The National Center for Biotechnology Information, The role of inflammation in epilepsy.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3378051/
3 Andrew H. Miller, et al. January 15, 2009.
The National Center for Biotechnology Information, Inflammation and Its Discontents: The Role of Cytokines in the Pathophysiology of Major Depression.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2680424/
4 S. Hu, et al. 2007.
The National Center for Biotechnology Information, Cytokine effects on glutamate uptake by human astrocytes.
https://www.ncbi.nlm.nih.gov/pubmed/10754403
5 Christof Grewer, et al. September 1, 2009.
The National Center for Biotechnology Information, Glutamate forward and reverse transport: From molecular mechanism to transporter-mediated release after ischemia.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2632779/
6 RB Laprairie, et al. October 2015.
The National Center for Biotechnology Information, Cannabidiol is a negative allosteric modulator of the cannabinoid CB1 receptor.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4621983/
7 LM Bornheim, et al. March 24, 1993.
The National Center for Biotechnology Information, Characterization of cannabidiol-mediated cytochrome P450 inactivation.
https://www.ncbi.nlm.nih.gov/pubmed/8466552
8 Giovanni Marsicano, et al. October 3, 2003.
The Realm of Caring Foundation, CB1 Cannabinoid Receptors and On-Demand Defense Against Excitotoxicity.
http://theroc.us/images/CB1%20Cannabinoid%20Receptors%20and%20On-Demand%20Defense%20Against%20Excitotoxicity.pdf
9 Matthew W Elmes, et al. February 9, 2015.
The National Center for Biotechnology Information, Fatty Acid-binding Proteins (FABPs) Are Intracellular Carriers for Δ9-Tetrahydrocannabinol (THC) and Cannabidiol (CBD).
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4423662/
10 The National Center for Biotechnology Information,
PubChem Compound Database.
https://pubchem.ncbi.nlm.nih.gov/compound/magnesium_ion
11 Maoxing Shen and Stanley A. Thayer. January 1999.
The National Center for Biotechnology Information, Delta9-tetrahydrocannabinol acts as a partial agonist to modulate glutamatergic synaptic transmission between rat hippocampal neurons in culture.
https://www.ncbi.nlm.nih.gov/pubmed/9882692
12 Duncan Ryan, et al. February 18, 2009.
The National Center for Biotechnology Information, Cannabidiol targets mitochondria to regulate intracellular Ca2+ levels.
https://www.ncbi.nlm.nih.gov/pubmed/19228959/
13 HR Ross, et al. April 7, 2008.
The National Center for Biotechnology Information, Inhibition of recombinant human T-type calcium channels by Delta9-tetrahydrocannabinol and cannabidiol.
https://www.ncbi.nlm.nih.gov/pubmed/18390906
14 Orrin Devinsky, et al. May 22, 2014.
The National Center for Biotechnology Information, Cannabidiol: Pharmacology and potential therapeutic role in epilepsy and other neuropsychiatric disorders.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4707667/
15 BE Porter and C Jacobson. December 2013.
The National Center for Biotechnology Information, Report of a parent survey of cannabidiol-enriched cannabis use in pediatric treatment-resistant epilepsy.
https://www.ncbi.nlm.nih.gov/pubmed/24237632
16 Jamar M. Cunha, et al. 1980.
The National Center for Biotechnology Information, Chronic administration of cannabidiol to healthy volunteers and epileptic patients.
https://www.ncbi.nlm.nih.gov/pubmed/7413719
17 Mayo Clinic,
Clinical trials of cannabidiol for epilepsy.
http://www.mayoclinic.org/medical-professionals/clinical-updates/neurosciences/clinical-trials-of-cannabidiol-for-epilepsy
18 American Epilepsy Society,
Three Studies Shed New Light on the Effectiveness of Cannabis in Epilepsy.
https://www.aesnet.org/sites/default/files/file_attach/AboutAES/PressReleases/cannabis%20release%20vfinal.pdf

Leave a Reply
You must be logged in to post a comment.